The New COVID-19 ‘Test to Treat’ Plan
During the first State of the Union Address of his term, President Biden announced a new national COVID-19 strategy as we begin year three of the pandemic. The ‘Test to Treat’ initiative increases preparedness plans and improves testing and immediate treatment access. In contrast to previous initiatives, Test to Treat allows those who test positive to immediately receive antiviral pills and other COVID-19 treatments at no cost.
Getting a test for COVID-19 following a known exposure or development symptoms is one of the most critical steps in managing the spread of the virus. With a Mira membership, you get at-home tests and access to low-cost lab tests, discounted prescriptions, and urgent and virtual care services. Sign up today!
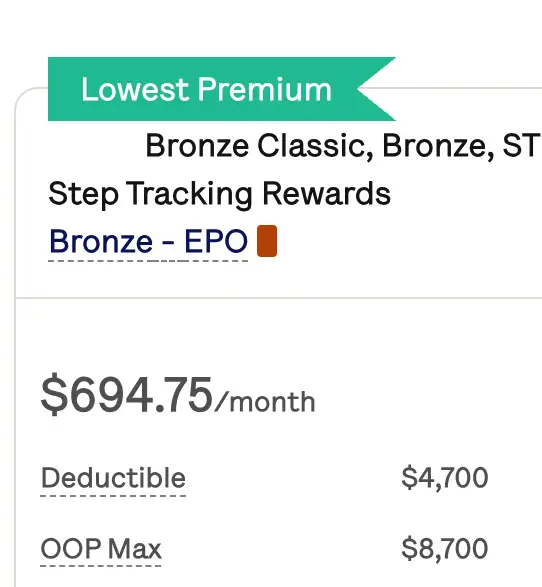
Get affordable doctor copay without paying insurance premiums
Join 39,000 people and get Mira, the best alternative to traditional insurance. Enroll and use immediately. Plans start at only $45/mo.
Originally from Houston, Texas, Alexandra is currently getting her Master's in Public Health with a health policy certificate at Columbia University. One of her life goals is to own her own art gallery!
