Should I Get a COVID Booster Shot This Fall?
According to the CDC, people ages 5 and up should receive a booster shot if it has been at least 2 months since their last COVID-19 vaccine dose. Studies have shown that the initial vaccine doses lose efficacy over time, and a booster dose will bolster immune responses from the original series of vaccines.
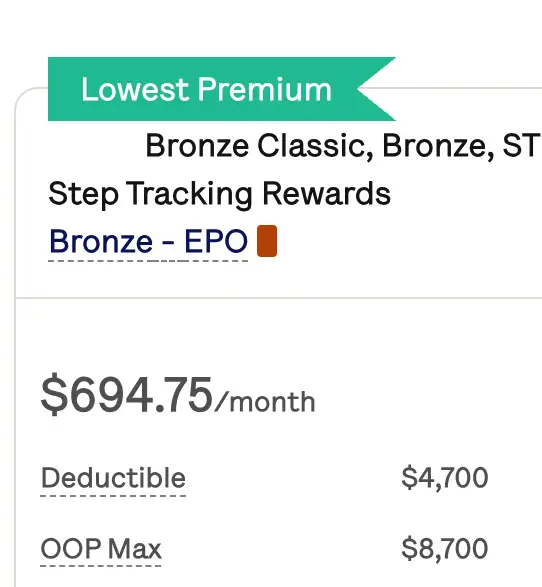
Get affordable doctor copay without paying insurance premiums
Join 39,000 people and get Mira, the best alternative to traditional insurance. Enroll and use immediately. Plans start at only $45/mo.
Shandra is a junior at Yale University, where she is studying biomedical engineering. Before joining Mira, Shandra wrote in the science and technology column of a local newspaper. Additionally, she has experience writing healthcare policy, covering topics such as the American Rescue Plan Act, abortion rights, and expansion of Medicare eligibility.
